

Our Science
Glucokinase Allosteric Modulators Dorzagliatin (HMS5552)
An Innovative Global First-in-Class Glucokinase Activator for Diabetes
Dorzagliatin (HMS5552) is an innovative global first-in-class oral antidiabetic therapy recently launched in China by Hua Medicine. As a novel glucokinase activator (GKA) with distinct chemical structure and unique mechanism of action (MoA), dorzagliatin has demonstrated its potential to restore the impaired glucose sensing function and achieve metabolic remodeling of glucose homeostasis in diabetic patients through repairing the defective glucokinase (GK), a critical glucose sensor in the glucose homeostasis. (Extended Reading: Dorzagliatin Tablet Approved for Marketing). Hua Medicine owns global rights to dorzagliatin and is working with strategic partners (Wuxi Apptec, Desano, Sinopharm) for commercialization in China.
Accumulating evidence from preclinical and clinical studies strongly indicate that dorzagliatin is able to repair and restore the impaired GK activity and function in T2D subjects, resulting in metabolic remodeling of glucose homeostasis. Dorzagliatin has demonstrated the ability to improve pancreatic function and insulin resistance with favorable safety and tolerability, thus holding great potential to stop or slow-down disease progression in T2D. Recently, we have demonstrated that dorzagliatin improves the impaired glucose stimulated GLP-1 secretion in obese diabetes patients and thus established the triple acting role of dorzagliatin in restoring the glucose homeostasis in diabetes patients. (Extended Reading: Glucokinase Activator Restores Glucose Sensitivity and Early-Phase Insulin Secretion in T2DM Patient—A Post Hoc Analysis of Dorzagliatin, SEED study: Dorzagliatin in drug-naïve patients with type 2 diabetes: a randomized, double-blind, placebo-controlled phase 3 trial, DAWN study: Dorzagliatin add-on therapy to metformin in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled phase 3 trial). This is for the first time that an oral antidiabetes agent demonstrated its effect on improvement of early phase insulin secretion and disposition index in large clinical trials and bring diabetes remission to drug naïve Type 2 Diabetes patients.
As a new generation GKA, dorzagliatin has been developed with a strategy to focus on increasing GK enzymatic efficiency (Vmax) at therapeutically effective concentrations, while maintaining GK's glucose sensing function (i.e., minimal change in the Hill coefficient nHill) without disruption of the glucose dependence of GK enzyme, an essential property for GK to act as a glucose sensor. This strategy has enabled us to achieve the ultimate goal of restoration of impaired glucose homeostasis in T2D patients. At the same time, extensive formulation optimization using state-of-art technologies has ensured the optimal distribution in key tissues and organs such as liver, pancreas and small intestines.
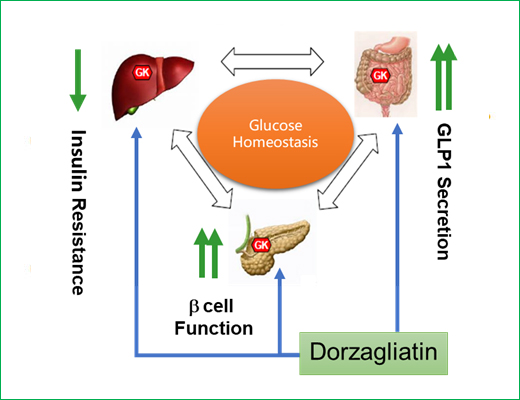
Impaired Glucose Homeostasis as a Central Hallmark of T2D
Glucose homeostasis is critical to human health, and is as vital as homeostasis for many other factors, such as water, electrolytes, oxygen, and body temperature. As an essential source of energy, glucose is necessary for maintaining normal functions of the nervous system as well as the immune system. Blood glucose concentrations for the majority of healthy individuals should be maintained within a narrow range of 4-6.5mM with a set point of 5 mM of glucose. Chronic deviation of blood glucose level from this range as a result of disrupted glucose homeostasis may lead to microvascular diseases, as well as diabetic nephropathy, diabetic retinopathy, diabetic feet, with increased risk for cardiovascular and cerebrovascular diseases.
T2D is a progressive disease with continuous decline in pancreatic β-cell functions regardless of intervention with currently available antidiabetic treatment, especially as monotherapy. Impaired glucose sensitivity in T2D patients results in pancreatic β-cell damage which in turn exacerbates blood glucose fluctuations, ultimately leading to disruption of glucose homeostasis with frequent complications as disease progresses. In T2D and PNDM patients, impaired GK expression, activity and function is one major cause for chronic hyperglycemia. Unfortunately, currently available anti-diabetes treatments, including insulin, are unable to address this underlying pathology in order to prevent the continued deterioration of pancreatic functions and control disease progression.
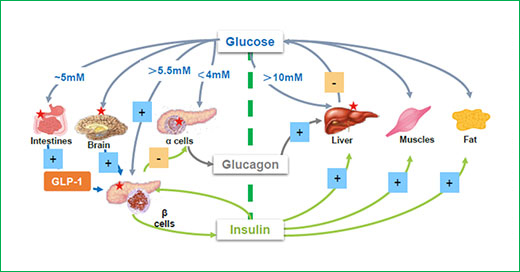
Glucose concentration sets the threshold for the secretion of hormones from endocrine cells which regulate glucose metabolism
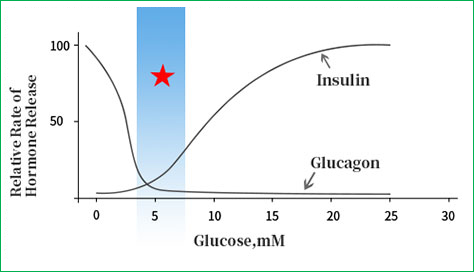
Relationship between blood glucose concentration and hormone release
As a Glucose Sensor, GK Plays a Central Role in Glucose Homeostasis
Similar to the room temperature sensor "thermostat", GK, as a glucose sensor in human, senses changes in blood glucose levels. GK automatically turns on the body's control mechanism to increase or decrease blood glucose in order to maintain the blood glucose level within the normal physiological range (4-6.5 mM). In pancreas, GK activity determines the glycemic threshold for triggering the K+-ATP channel–mediated release of insulin or glucagon, two key hormones in the regulation of glucose homeostasis.
20 Years of Development of Glucokinase Activators
Since the initial publication of the pharmacological basis for GKA in 2001, research and development in this area has flourished. So far, more than 100 patents on GKA have been published, and over 20 GKAs have entered into clinical trials, among which Dorzagliatin is the most advanced one launched in China to improve the impaired glucose homeostasis in T2D patients.















